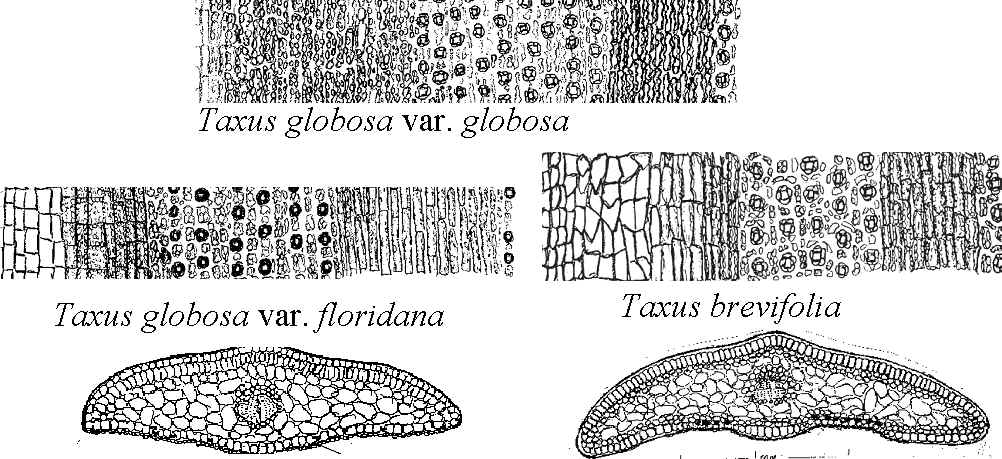
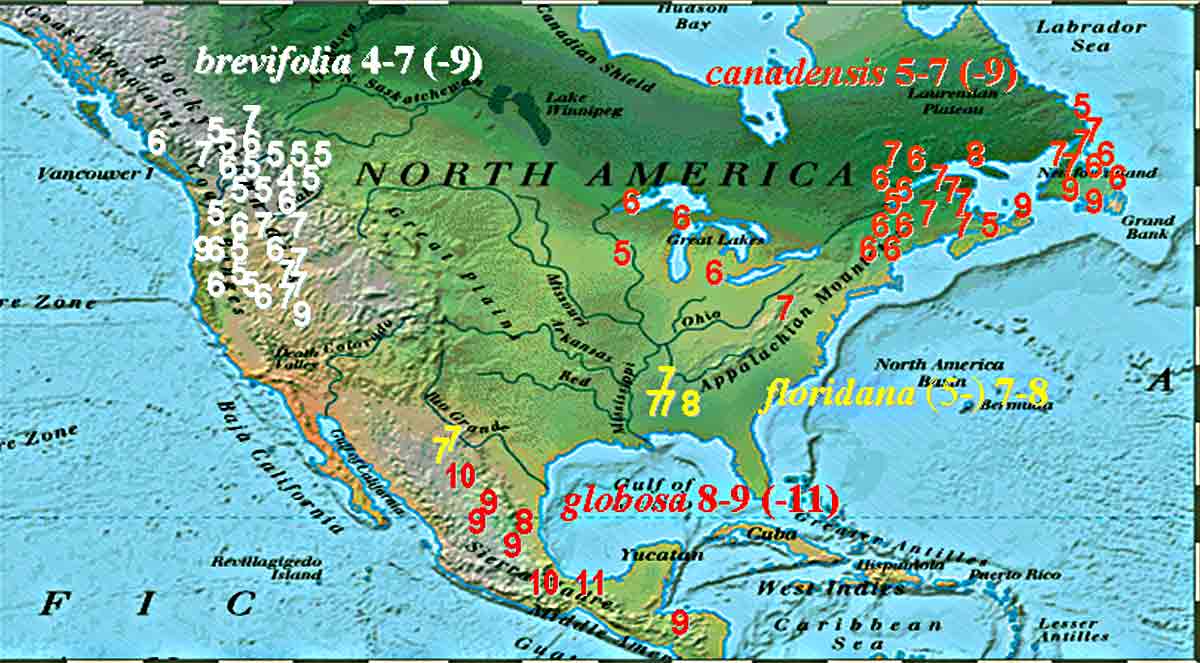
Leaf cross sections and abaxial epidermal surface sections of the Taxus wallichiana Subgroup in North America and map showing number of stomata rows for the species, including also T. canadensis The Taxus wallichiana Subgroup in North America is characterized by leaves having tall rectangular to nearly quadrangular epidermal cells as seen in cross section (illus. above map), stomata in 12 rows/band, and a papillose abaxial midrib (top two rows of illus.). There are two species, Taxus brevifolia, subdivided into four varieties, var. brevifolia, var. klamathensis, var. polychaeta, and var. reptaneta, and T. globosa subdivided into two varieties, var. floridana and var. globosa. The two species are distinguished by number of stomata rows in a stomata band, 4–7 (-9) in T. brevifolia, and 7–11 in T. globosa as plotted on the map above from plant specimens in herbaria and collected in the field, and by the shape of the marginal cells that border stomata bands, appearing ± rectangular in T. globosa, irregular and inflated in T. brevifolia. Taxus canadensis, which also occurs in the Mediterranean Region (Spjut , is classified in the Taxus cuspidata Alliance (Spjut 2007b) of the Taxus baccata Group. It is recognized by the absence of papillae across the midrib and marginal cells on the abaxial surface (Spjut 1992, 1993, 2000, 2007b). The numbers preceding the species names are from Spjut (2007b) in which the Wallichiana Group includes species from Asia; all are arranged alphabetically by species within the group.
3. Taxus brevifolia Nutt. Detailed description is provided for each four varieties under separate web pages. The four varieties of T. brevifolia are distinguished by differences in the morphology of ovuliferous shoots and by the habit of the plant as follows:
var.
brevifolia: trees with branches
spiraled all around the bole, regenerating by adventitious shoots var. klamathensis: trees regenerate branches on bole and regenerate clonal trees by adventitious shoots arising from long lateral roots while the main trees still grows. Plants self-pruning when growing in open areas. Ovuliferous shoots similar to var. brevifolia, or to var. polychaeta in producing more than one ovule (up to 5 ovules) but differing in the ovular (secondary) shoot having three pairs of decussate scales as generally found in Taxus. var. polychaeta: distinguished by cylindrical (worm-like) ovuliferous shoots.
var.
reptaneta:
shrubs reproducing by layering branches; main branches ascending,
secondary branches | ||||||||||
|
7. Taxus
globosa Schlectendahl, Linnaea 12: 496 (1838), no
specimens cited. Lectotype (designated by Spjut 2007b): Mexico, Hidalgo: Real del Monte, Cerro de las
Nabajas, [Sep] 1837 Ehrenberg s.n. K Mesoamerican yew. Distribution: El Salvador and Honduras to Florida. 7a. Var. globosa. Low spreading shrub or tree, to 40 m tall, bole to 40 cm diam.; bud-scales loosely persistent, 2–3 seriate, brownish, 1.5 (-2) mm long, chartaceous, dull, lower scales ovate, upper narrower, plane to slightly concave, carinate towards apex, or cuspidate. Leaves spreading as in T. brevifolia, overlapping in two-ranks, linear-falcate, 1.5–2.2 (-3.5) cm long, 1.5–2.2 (-2.5) mm wide, 200–350 µm thick, pale to dark green and slightly convex above to a rounded midrib that is slightly channeled at base, yellowish or pale green and slightly concave below to a slightly rounded midrib, plane to revolute near margins, acute to apex. Upper (abaxial) epidermal cells isodiametric to slightly elliptical in transverse section, or slightly taller than wide, 20–25 (-40) µm tall, 20–30 (-45) µm wide; lower epidermal cells similar to upper in transverse section, fusiform to rectangular near margins, slightly inflated, narrower and more incrassate towards stomata bands, mostly 3–10×+ l/w, longer on midrib, papillose to 3–4 rows from margins; papillae conspicuous in 2–3 alternate rows on each cell, positioned submarginally and marginally (along cell walls); stomata (7-) 8–9 (-11) rows/band. Male cones not studied; aril red, maturing in the fall. Mesoamerican yew. Distribution: montane cloud forests above 2,000 m, S Mexico (Veracruz, Hidalgo, Mexico, Oaxaca), Guatemala (Baja Verapaz, El Progreso [Sierra de las Minas, Volcán de Santa Luisa], Zacapa, Volcán Gemelos, Monte Virgen] Huehuetenango [Sierra de los Cuchumatanes, Cerro Cananá]) to British Honduras (Merrendón, San Idalfonso, Omoa, Montecillos, Opalaca, Congolón) and El Salvador. Representative Specimens—Mexico: Trinidad Iron Works, 24 May 1904, Pringle s.n. (US). Tamaulipas: from El Cielo to Ojo de los Indios and the Parque above Gómez Farías, A. J. Sharp 52112 (GH). Veracruz to Orizaba, 1851, Meisner s.n., Herb. Gordon (K). Hidalgo: barranca below Honey Station, Pringle s.n. (BH); Real del Monte, Hartweg 438 (BM). Guatemala: Dept. El Progreso: hills N of Finca Piamonte, 2400–3333 m, Steyermark 43487 (F); W end of Sierra Las Minas, Baja Verapaz, 6500 ft., Sharp 45247 (F). Honduras: Cerro Sta. Barbara, steep rocky slopes, 2750 m, tree 30–50 ft, usually of low spreading, or even semi-procumbent habit, 10–12" diam. at base, Armour & Chable 6083 (US). El Salvador: Dept. Chalatenango, Summit to Los Esesmiles, 14º20'N, 89º09'W, dense cloud forest, 2670 m, Tucker 1073 (BH, PH, US). This species is recognized by the abaxial leaf epidermis having relatively narrow wavy-walled cells. Specimens from northern Mexico are not easily distinguished from those in Florida, thus, I conclude that the Florida yew also occurs in Mexico, and that differences between the two varieties are relatively minor compared greater variation in their Asian relatives. The Florida yew is also difficult to distinguish from related species in SE Asia, particularly T florinii in Sichuan and Yunnan, and T. phytonii in the Philippines, Taiwan, and Yunnan their epidermal cells differ in being slightly inflated near margins, and by the slightly elliptical shape in T-section. 7b. Taxus globosa var. floridana (Nutt. ex Chapman), Spjut, J. Bot. Res. Inst. Texas 1(1): 224. Taxus floridana Nuttall, Chapman, Fl. South. U.S. 436 (1860), no specimens cited; T. baccata ssp. floridana (Nutt.) Pilger, Planzenreich 4(5): 113 (1903); T. canadensis Marshall var. floridana (Nutt. ex Chapman) Silba, Phytologia Mem. 7: 72 (1984). Type. U.S.A. Florida: near Aspalaga, 1833, Croom s.n.; lectotype (designated by Spjut 2007b): PH! (with label in Nuttall handwriting). Specimens referred to as T. croomii, which were obtained by Chapman, are also original material, but are not isolectotypes as earlier noted on this site. Tree or shrub, 6–10 m tall, bole to 40 cm diam. Leaves mostly (1.0-) 2.0–3.0 cm long, 0.3–1.5 (-2.2) mm wide, 200–350 µm thick, often glossy dark green and convex above to a rounded midrib, yellowish or pale green and concave below to a rounded midrib, recurved near margins. Epidermal cells nearly isodiametric to nearly quadrangular in transverse section, similar in height on both surfaces, 25–40 µm diam., those on abaxial surface uniformly rectangular, thick-walled, larger or slightly inflated or irregular in shape in up to 6 rows nearest stomata bands, mostly 1–5× l/w near margins, more than 10× l/w on midrib; papillae lacking on 5 or more cells across margins, mostly concrescent, globose to cylindrical, in 2–3 linear rows on each cell, positioned marginally, along cell walls, prominent in stomata bands, less developed on midrib; stomata (5-) 6–8 rows. Male cones numerous on young branchlets, scales 3-seriate, microsporophylls ca. 8, each with ca. 6 cucullate microsporangia. Female cones on 1st yr branchlets, the scales decussate in 3–4 ranks; aril red, Sep.-Oct.; seed conical, 4-angled. Florida yew. Rare, W Florida: Apalachicola River; Chamaecyparis swamp ca. 8 mi SE of Bristol) (http://www.floridata.com/ref/T/taxu_flo.cfm); N Mexico (Nuevo Leon, Tamaulipas). Representative Specimens—Mexico—Veracruz: 2200 m, Hernandez 01459 (BM). Tamaulipas: Sierra Madre Oriental, ca. 30 mi S of Monterrey, C. H. & M.T. Mueller 1337 (BM, PH); Tamaulipas/Nuevo Leon, 24º99'N; 100º5'W, 1690 m, Meyer & Rogers 2746 (BM). U.S.A.—Florida: near Bristol, Mohr s.n. (PH), ex Canby Herb. (PH); Rock Bluff, limestone, Blanton 7050 (PH), Wherry s.n. (PH). Cultivation. University of Florida, Gainesville, hedge like shrub outside campus building, Gilliland & Spjut 12172 (wba); fresh specimen without collector or locality, Snader s.n. (National Cancer Institute). Other material submitted by Phyton (Ithaca, NY). This Florida variety differs from the typical variety by the broader marginal area of rectangular epidermal cells on the ventral (abaxial) surface of the leaf, and by the slightly lower stature in development of papillae. The Florida yew is exceptional in occurring at relatively low elevations in seasonally hot humid forests and also having leaves with a papillose midrib and angular (isodiametric) cells. These features are usually found in yew species that grow above 2000 m. At lower elevations, as seen for yews in China, yew leaves generally lack papillae on the abaxial midrib, and their epidermal cells in T-section appear more rounded elliptical than angular. The Florida yew is clearly related to the Mesoamerican yew. It may have found refuge at lower elevations along the Gulf as glaciers advanced during the Pleistocene. Compared to a wide range in differences seen in Asian yew, the Florida yew evidently has not had much time to differentiate itself from the Mesoamerican yew.
|
||||||||||
|
Spjut, R. W. 2014. A new variety of Taxus brevifolia from the Pacific Northwest of North America. Abstract submited to the IV Internacional Yew Workshop: Management, conservation and culture of the yew forests in Mediterranean forest ecosystems. Paratge Natural d’Interès Nacional de Poblet October, 23-25 2014. Spjut, R. W. 2007b. Taxonomy and nomenclature of Taxus. J. Bot. Res. Inst. Texas 1(1): 203–289.
Spjut, R. W. 2000,
The morphological relationships of Taxus canadensis (Taxaceae) in
North America and
Eurasia. http://www.2000.botanyconference.org/section13/abstracts/28.shtml Spjut, R. W. 1993. Reliable morphological characters for distinguishing species of Taxus (Abstract). In International yew resource conference. Yew (Taxus) conservation biology and interactions. Berkeley, CA. Pp. 39–40. Spjut, R. W. 1992. A taxonomic key to the species of Taxus. NCI Workshop on Taxus, Taxol, and Taxotere, Rockville, MD (Abstract only).
|